*with 6 hours of free training*

Sign up for a free 30-day trial of ryze clinical trial software
Complete this form to request your FREE software trial. We’re including 6 hours of free training on the ryze platform, to help you get the most from your trial. There’s no obligation, so what are you waiting for – complete the form now and let’s see how better off you could be with ryze!
Easily find, share, standardize and reuse content
See how CRFs look before building your EDC
Shorter CRF approvals
Build your full study from standards in ryze
See trial data from 1st patient in
Easily comply with CDISC
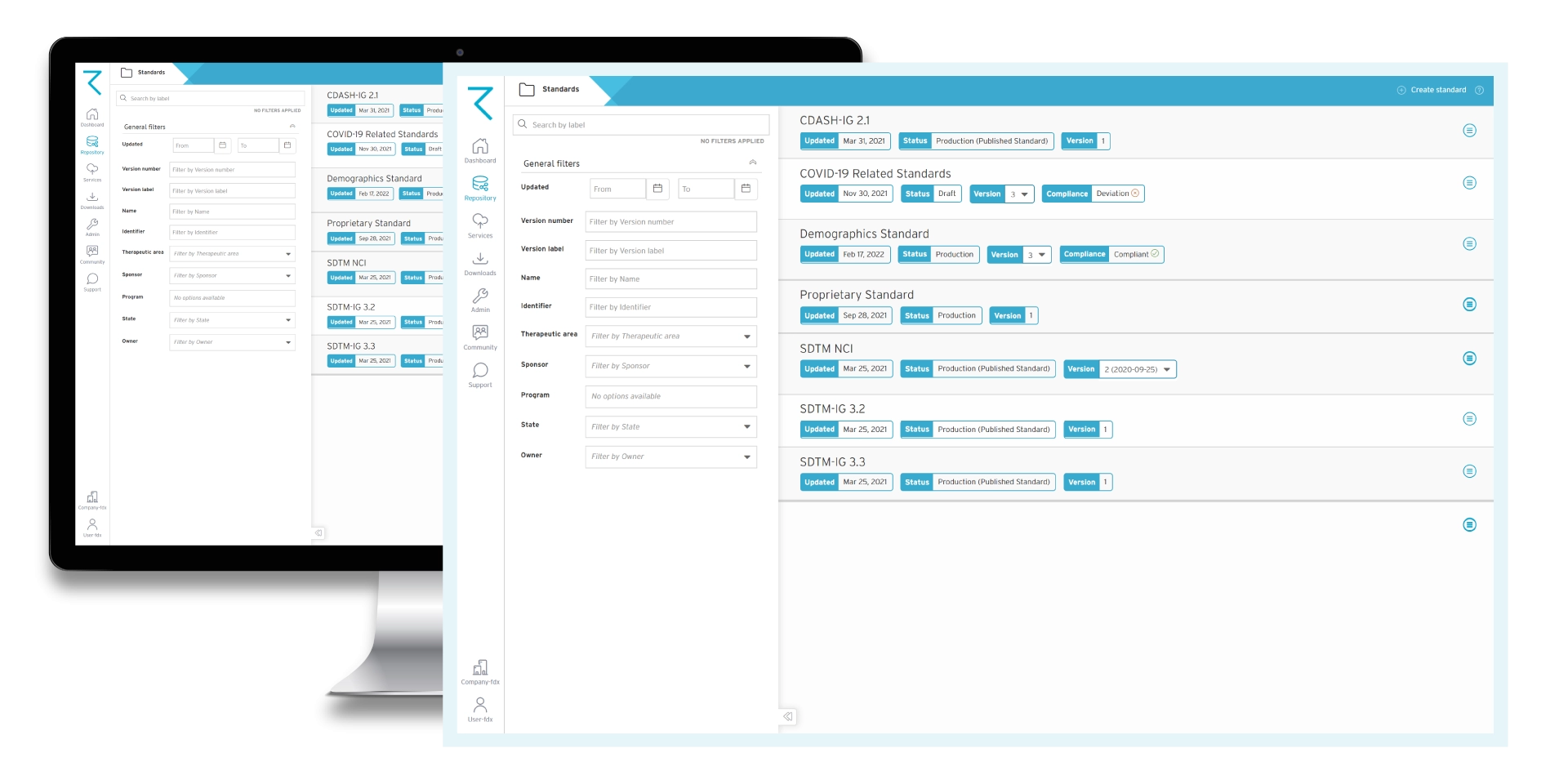
Features of ryze Clinical MDR & automation suite
Organizations using our clinical trial software











What people say about us




VAT No. GB 671715037 | Company number SC159080